An Accurate and Reliable Method for Measuring Colonic Transit Time
Radiopaque marker studies are an effective method for measuring colonic transit time (CTT). The results obtained from this technique are highly dependable, but the accuracy of the results may vary depending on the specific technique used. Therefore, it´s important to choose the right technique to ensure the most accurate results (Keller).
The Transit-Pellets method is a validated method and provides precise measurement of total and segmental colonic transit time. This method involves the ingestion of Transit-Pellets radiopaque markers in multiple capsules over a set period of time, followed by a single abdominal X-ray on day seven (Hasse, Aziz).
In contrast, a single capsule technique is a screening method and involves the ingestion of markers in a single capsule on a specific day, followed by multiple abdominal X-rays. This method is time-consuming and exposes the patient to higher levels of radiation. It is also qualitative, meaning that it does not provide numerical values for CTT. Instead, it is used to determine normal from slow transit (Kim, Keighley, Popescu).
Using the Transit-Pellets method and Transit-Pellets radiopaque markers exposes the patient to lower levels of radiation, while still providing numerical values for CTT.
Transit-Pellets Device Description
Innehåll här? Se kommentar i Dianas dokument.
Measurement of Colonic Transit Time with the Transit-Pellets Method
The Transit-Pellets method, also known as Abrahamsson´s Method, offers a valuable alternative for measuring colonic transit time (CTT) in patients, as it allows for quantification of CTT while minimizing exposure to radiation. This method is unique in that it is the only single X-ray approach that addresses a range of practical considerations, such as gender-specific differences, both rapid and slow CTT, and the ability to locate dysfunction in various segments of the colon. By achieving an equilibrium between daily marker input and output, this method provides accurate and reliable numerical values for CTT.
The Transit-Pellets method and the Transit-Pellets device can be used to:
- Measure rapid, normal, and slow colonic transit1,2
- Differentiate between slow transit and normal transit constipation3,4,5
- Identify segmental colon dysfunction in patients with constipation 3
- Differentiate between normal and rapid transit diarrhea6
- Identify treatment effects in patients with chronic constipation7
Gender Difference in Colonic Transit
Healthy females generally tend to have a notably slower intestinal transit compared to males (Sadik, Disney). In light of this, it is essential to take into account factors such as gender in the selection of a method for measuring colonic transit time. The Transit-Pellets method is designed to provide accurate measurement of colonic transit time, taking into account gender-specific variations by utilizing robust reference values (Hasse, Sadik).
Accurate Measurement of Rapid Colonic Transit
The Transit-Pellets method and the Transit-Pellets device allows for the measurement of rapid transit, which is of significance in certain populations experiencing chronic diarrhea. This is possible due to the short interval of 12 hours between the final marker intake on the sixth day and the subsequent X-ray taken on the seventh day. This schedule serves as the foundation for determining the lower limit of normal transit (the 5 th percentile) and identifying cases of rapid transit (Sadik).
Preparing for the Colonic Transit Test with Transit-Pellets Radiopaque Markers
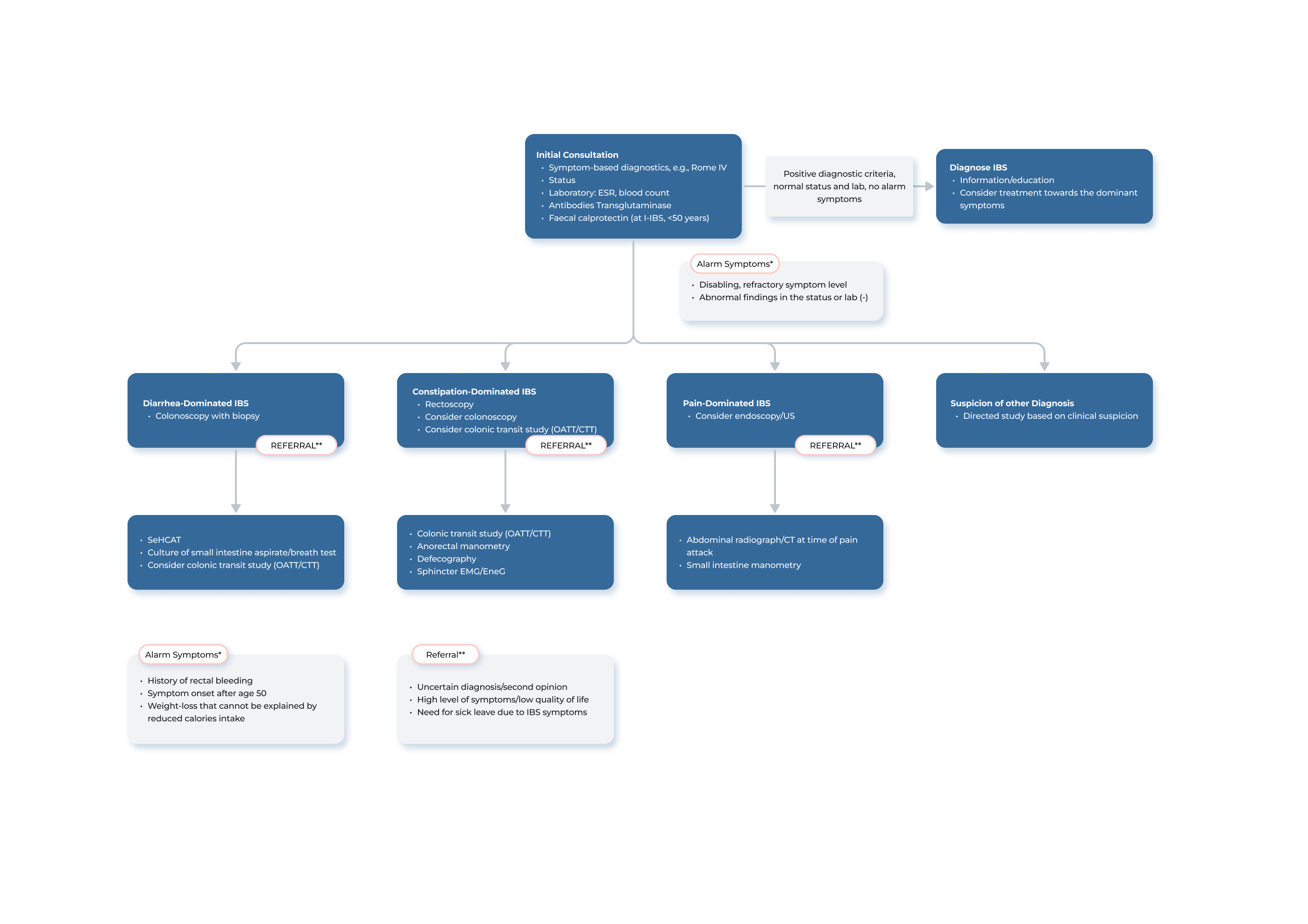
A radiopaque marker study with Transit-Pellets is recommended for clinical evaluation of colonic transit time (CTT) in subjects with constipation and irritable bowel syndrome (IBS). The test is indicated to differentiate between normal and slow transit, to assess segmental transit times in patients with delayed total colon transit and sometimes to assists in the evaluation of unexplained chronic diarrhea.
The Transit-Pellets radiopaque markers are prescribed by a physician for use by the patient in the home setting. It is imperative that patients receive adequate instruction regarding the proper handling of the Transit-Pellets device to ensure safe usage.
The referral for the Transit-Pellets diagnostic procedure may be initiated by a primary care physician or other physician with expertise in treating patients with gastrointestinal conditions. In instances where the case presents complexity or uncertainty, the referral may be directed directly to a gastroenterology specialist for further evaluation and assessment.
Transit studies primarily address the question, “does the patient have normal or slow transit?”. The assessment of segmental transit time is of paramount importance when evaluating patients with delayed transit. In order to receive a thorough report from the radiology department, it may be advisable to include a request for the identification of any specific segments of the colon that exhibit delays in transit.
In the standard protocol, the X-ray department assumes responsibility for providing instruction and overseeing the use of the Transit-Pellets device. The provision of clear instruction and information to the patient is a critical component of the diagnostic procedure. In cases where assessment proves to be challenging, consultation with a gastroenterologist or a specialized gastrosurgeon may be sought for guidance.
For further guidance on the appropriate utilization of the technique and Transit-Pellets device, kindly refer to the Transit-Pellets Clinical Instruction for Use. Please be aware that the Clinical Instruction for Use is subject to change. The most current version is always available on this site.
Prescription pad, X-ray and Order Form.
From Diet, Laxatives and Lifestyle Modifications to Colonic Transit Studies
The principles for determining the timing of colonic transit time measurements in relation to treatment steps may vary based on the specific center and the individual physician´s experience. In general, the initial management approach for constipation includes focusing on diet and lifestyle modifications, as well as utilizing standard over-the-counter laxatives such as bulking agents and non-absorbable carbohydrates. If these measures are ineffective, it is common to proceed with a colonic transit study with Transit-Pellets radiopaque markers. Some gastroenterologists may also try additional treatment methods, such as the use of saline laxatives or motility-stimulating drugs, before arranging for a colonic transit test.
(flowchart?)
Record Bowel Movement with the Bristol Stool Chart
The Bristol Stool Chart was developed in 1997 as a clinical assessment tool (Lewis). It is commonly utilized to describe the forms and consistency of stools, as well as to diagnose conditions such as constipation, diarrhea, and irritable bowel syndrome.
When scheduled to take a colonic transit test with Transit-Pellets radiopaque markers, it is recommended that patients keep a record of their bowel movements throughout the week. The Bristol Stool Form Scale (BSFS) can be used to evaluate the consistency of their stools on a seven-point scale and may assist in this process. By comparing the patient´s reported symptoms, bowel movement frequency, and the test results, the physician can gain a better understanding of the patient´s condition and the test results in conjunction with the diary can be useful for clinical practice.
Instructions on Timing and Administration of Transit-Pellets Capsules
The procedure for the colonic transit test using Transit-Pellets radiopaque markers involves the patient ingesting ten markers for six consecutive days. The recommended time frame between the initial marker consumption and the X-ray examination is approximately 144 hours (Hasse).
Advise the patient to follow the Transit-Pellets capsule regimen as detailed below: One capsule per day is to be consumed from day one through day five. On day six, the patient should consume one capsule labeled “6:1” in the morning and one capsule labeled “6:2” in the evening. Emphasize the importance of adherence to this schedule to the patient in order to obtain accurate results.
It´s also important that the capsules are taken at around the same time each day. For example, if the first capsule on day one is consumed at 8:00 am, the capsules on days two, three, four and five should also be taken around that same time. Additionally, on day six, the capsule labeled “6:1” should be consumed at approximately 8:00 am, while the capsule labeled “6:2” should be consumed around 8:00 pm the same day approximately 12 hours prior to the X-ray on day seven.
For patients who may have difficulty swallowing capsules, an alternative method of ingestion can be employed. The contents of the capsules can be emptied into soft foods such as applesauce or yogurt and consumed in that form. It is important to note that the capsules and markers should not be chewed.
Schedule a Plain Abdominal Radiograph or a Fluoroscopy
The X-ray or fluoroscopy on day seven should be scheduled to take place about 12 hours after the patient´s last dose on the sixth day. Therefore, the time of the X-ray determines when the patient should take the Transit-Pellets capsules, both in terms of the day they should begin and the specific time they should take them. The following table provides a helpful reference:
TABELL?
Adherence to the prescribed marker ingestion protocol is crucial for accurate results. However, a deviation of up to an hour in the timing of marker ingestion is permissible, but it is not recommended for clinics to routinely have patients ingest markers in the morning and then perform radiographic examinations later in the day, as this could lead to unreliable results.
The Importance of Maintaining Normal Dietary and Fluid Habits During the Test
As the patient ingests the Transit-Pellets markers during the course of the week, it is recommended that they maintain their normal dietary and fluid intake habits. This is to ensure that the results obtained are not influenced by any external factors.
Avoiding Laxatives During the Test: Importance for Accurate Results
For the duration of the test, it is important for patients to refrain from using laxatives. While it may not always be feasible to avoid motility altering drugs, it is important to consider their potential impact when interpreting the test results (Keller).
Transit-Pellets Instruction for Use
Transit-Pellets come with an Instruction for Use (IFU) that is available in up to 20 languages. You will find it in the footer menu “Downloads” or you can enter it directly via THIS LINK. The IFU provides important instructions on the proper use of Transit-Pellets and should be thoroughly reviewed to ensure safe and effective use of the device.
What to Do if a Patient Misses a Dose?
If a patient has missed a dose of Transit-Pellets radiopaque markers, it is not recommended to compensate by taking a larger number of markers on a subsequent day. This is because the order of the patient´s transit time cannot be accurately determined, and it is therefore not possible to accurately assess the total and segmental colonic transit time using the markers. If a patient has missed the prescribed intake of markers on two occasions, it may be advisable to try again at a later time to ensure accurate results.
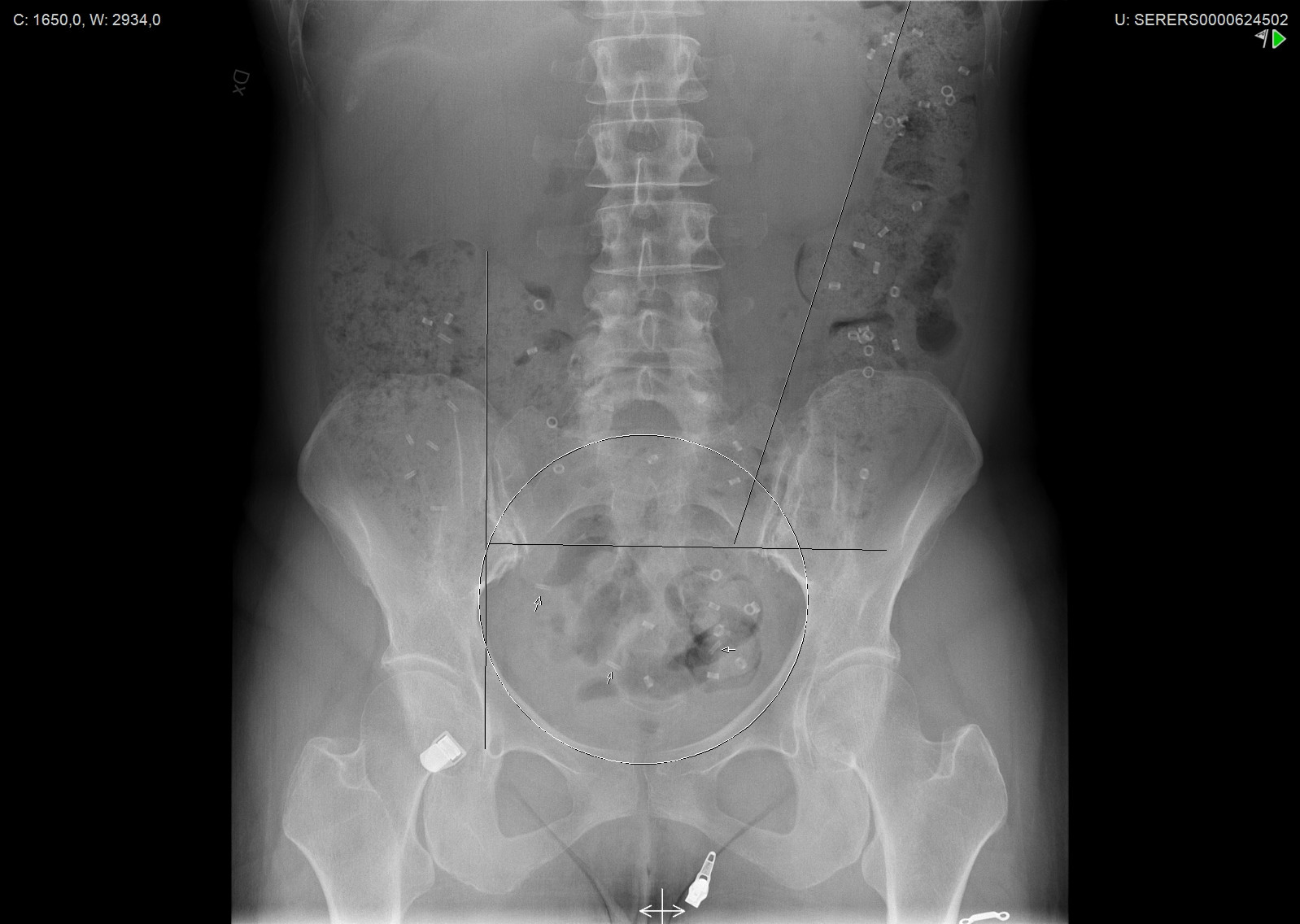
Analysis of the Abdominal Radiograph
The analysis of the abdominal radiograph taken on the seventh day involves the visual inspection of the X-ray to count the remaining Transit-Pellets radiopaque markers in the colon, as well as the identification of their distribution across the different segments of the colon.
By using multiple markers over a longer period of time, it eliminates the need to track specific markers from day to day and provides a more comprehensive and accurate measurement of the colonic transit.
It is routine to initially evaluate the overall colonic transit, which is deemed the primary indicator. If the overall transit is abnormal, it is essential to further assess individual segments to identify the location of any delays within the colon.
For calculation of segmental transit, four segments are used; cecum and colon ascendens including the right flexure, the transverse part, colon descendens including the left flexure, and sigmoid colon-rectum (see Figure X).
Iqi, här kommer vi behöva skapa en snygg bild liknande nedan.
Some clinicians divide the colon into three regions based on bony landmarks (Ahran): the right colon, left colon, and sigmoid colon-rectum regions. Both approaches are medically equivalent and it´s up to the individual clinic to choose the principle they are most comfortable with.
Calculation of Total and Segmental Transit Time
Colonic transit is calculated as the mean oro-anal transit time (OATT, mouth-to-anus) for the daily marker doses swallowed. Because the colonic transit constitutes the main part of the mouth-to-anus transit time, OATT is used as a measure of colonic transit. The transit time is equivalent to the number of daily marker doses retained.
When conducting a Transit-Pellets test, it is typically not necessary to differentiate between ring-formed and tube-formed markers. Each marker should be considered as one unit, regardless of its shape. With ten markers per day, each marker is equivalent to 0.1 days (or 2.4 hours).
The formula is straightforward, it involves dividing the number of remaining markers by the daily dose, which in this case is 10 markers. Clinics can use the formula M/10 to determine both total and segmental transit time in days. Some clinics may prefer to present the results in hours, in which case the formula Mx2.4 can be used.
If for example a total of 35 markers are retained, the OATT is determined as 3.5 days (or 84 hours). In case of retention of a total of 15 markers in a specific segment, the OATT is determined as 1.5 days (or 36 hours) in that segment.
Why Tube-Formed Markers on Day Six?
The use of tube-form markers on the sixth day serves a specific purpose in certain cases. In patients with slow transit, it may be difficult to accurately locate the cecum. The use of tube-form markers in these cases can help in determining the separate transit times for the various colonic segments. As the tube-formed markers are the last markers to be ingested, their presence in the colon can indicate the location of the cecum. Additionally, a redundant sigmoid colon may overlap with the right colon, while a large cecum can crossover to the rectosigmoid area, the use of tube-formed
markers can assist radiologist in identifying the localization of the markers in the correct colonic segment.
The tube-formed markers ingested on day six can also provide valuable information in cases of rapid transit which is of significance in certain populations experiencing chronic diarrhea (Sadik).
It is also worth noting that if at least six markers from the sixth day have not reached the colon by the seventh day, it may be an indication of slow transit in the small intestine, which is a common occurrence in patients with constipation.
Why 22% Barium Sulphate?
International experts recommend using a 20-25% proportion of heavy barium sulphate in Transit- Pellet markers to accurately measure colonic transit time (Cummings). Using a higher proportion of barium, such as 40%, can lead to inaccurate results and a specific gravity above the recommended range of 1.2-1.7 for colonic contents.
Interpretation
The total number of remaining markers in the colon determines if the colonic transit is delayed or not. A value above the upper reference value (95 th percentile) indicates a delay in transit, whereas a value below the lower reference value (5 th percentile) indicates an abnormally rapid transit.
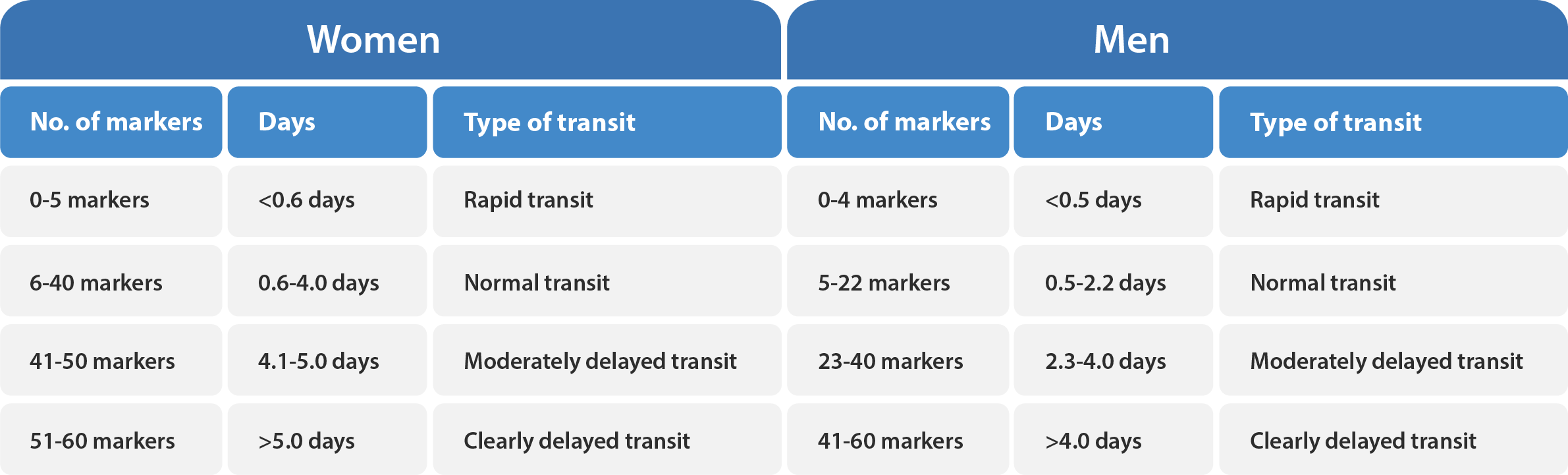
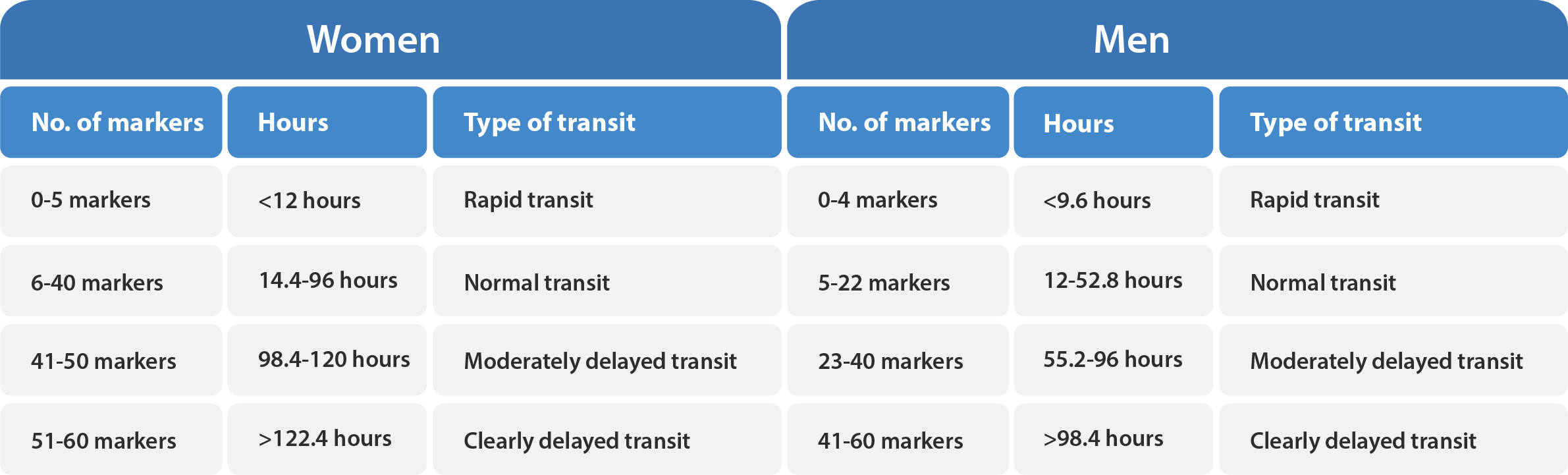
For healthy women, the upper reference value for colonic transit time is 4.0 days (40 markers), and a value between 4.1 and 5.0 days is considered moderately delayed transit, while a value greater than 5.0 days is considered clearly delayed transit. On the other hand, healthy men have a more rapid mean transit time than healthy women. The upper reference value for men is 2.2 days (22 markers). Please refer to Table X and X below for additional guidance.
If a patient has a value below the lower reference value (0.6 days or 6 markers for women and 0.5 days or 5 markers for men), this is considered to be an abnormally rapid colonic transit.
Additionally, the distribution of markers in the various colonic segments can provide information about the type of delay. It is, however, important to note that healthy individuals may have a transit value in a few segments that approaches the upper reference value, but not in all segments at the same time, as indicated by the reference value for the total transit time.
A numerical transit time value can be given if the number of retained markers is in the range 3–55 markers. Thus, at least half a daily dose should be excreted and at least half of the evening dose on day six must be retained. If the number of retained markers is only 0–2, the transit time is less than 0.3 days. If 56–60 markers are retained, the transit time is more than 5.5 days. This is an indication that equilibrium has not been reached yet.
The tables below provide a reference for the assessment of total and segmental colonic transit.
These values may be used as a guide for evaluation.
For clinics that prefer to obtain results in hours, please click here.
(tabellerna som presenterar resultatet i timmar ploppar upp. Jag vill få bort de eftersom det blir lite rörigt att visa både in days and hours)
The Importance of Segmental Transit Time Measurement
In a clinical practice, it is standard practice to initially assess the overall value, which is considered the most important parameter. Should the overall value be elevated, it is imperative to evaluate individual segments to ascertain the location of any delays within the colon.
The measurement of segmental transit time is of significant interest in evaluating patients with delayed transit. This is because it provides an indication of which segment of the colon may be affected. For example, it can indicate if the dysfunction is localized in the recto-sigmoid area, or if it is more widespread and affects the left colon or even the entire colon, as seen in cases of colonic inertia (FAQ).
It is also recommended that the segmental transit time be calculated in patients with both rapid transit and diarrhea. For example, if a patient with diarrhea has a recto-sigmoid segmental transit time that falls within the upper range of normal transit, it can effectively rule out the possibility of bile acid diarrhea (Sadik, ref. 4).
Applications of the Results
Upon completion of the CTT measurement, it is important to review the results with the patient and present the obtained transit profile. The transit profile should in general be interpreted in the context of the patient´s reported symptoms.
The following is a suggested approach for utilizing the results of a colonic transit test with Transit-Pellets radiopaque markers. It is important to note that the ultimate decision regarding the interpretation and application of these results, as well as the course of subsequent treatment, should be made by the treating physician in consultation with the patient.
- If the results indicate that the overall transit is normal, patient education, dietary guidance, and/or osmotic laxatives are often sufficient. If dyssynergic defecation is present, biofeedback therapy may be recommended
- If the results show that the colonic transit is delayed, intensification of constipation therapy, including adjustments to laxative treatment and the use of motility-stimulating medications, may be necessary
- In slow-transit constipation, radiopaque markers are typically retained throughout the entire colon, while concentration of markers in the cecum and ascending colon, may indicate colonic inertia
- If the patient experiences severe constipation but the transit time is completely normal, it is possible that altered sensitivity, such as irritable bowel syndrome (IBS), is present and therapy should be tailored accordingly
- If a patient´s colonic transit test reveals a delayed transit time in the recto-sigmoid region, it may be indicative of an outlet obstruction disorder. In such cases, further investigation may be
required, such as balloon expulsion test or ano-rectal manometry may be needed to determine the functional or structural cause of evacuatory dysfunction - If a patient is experiencing chronic diarrhea and the results shows rapid transit in the distal colon, it may be indicative of bile acid diarrhea
For further information regarding Professor Hasse Abrahamsson´s perspective on crucial factors to take into account when conducting a colonic transit test utilizing the Transit-Pellets method and Transit-Pellets radiopaque markers, we recommended visiting the section Why assess Colonic Transit Time (CTT) with the Transit-Pellet single X-ray method?
The information provided on this website is intended for general information purposes only. It is based on a compilation of various articles, literature and previous material written by Prof. Hasse Abrahamsson. The final decision regarding the use of the Transit-Pellets device and the interpretation of the results rests with the physician. Medifactia does not provide medical services or advice and the information on this website should not be considered as medical advice. It is important to consult with a qualified healthcare professional for any specific medical needs or questions.
References
Articles
Keller, J., Bassotti, G., Clarke, J., et al. (2018). EXPERT CONSENCUS DOCUMENT. Advances in the diagnosis and classification of gastric and intestinal motility disorders [electronic version]. Nat Rev Gastroenterol Hepatol., Vol. 15(5), 291-308.
Aziz, I., Whitehead, W.E., Palsson, O., Törnblom, H. & Simrén, M. (2020). Expert Review of Gastroenterology & Hepatology. An approach to the diagnosis and management of Rome IV functional disorders of chronic constipation [electronic version]. Taylor & Francis Group, Vol. no. 14, 1-8.
Kim, E.R. & Rhee, P-L. (2012). How to Interpret a Functional or Motility Test – Colon Transit Study. J Neurogastroenterol Motil, Vol. 18(1), 94-99.
Lewis, S.J. & Heaton, K.W. (1997). Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol., Vol. 32(9), 920-4
Arhan P., Devrode G., Jehannin B., et al (1981). Segmental colonic transit time. Dis Colon Rectum, Vol. 24(8):625-629.
Popescu, M. & Mutalib, M (2021). Bowel transit studies in children: evidence base, role and practicalities. Frontline Gastroenterol., Vol. 13(2), 152-159.
Cummings, J.H., Jenkins, D.J.A., Wiggins, H.S. (1976). Measurement of the mean transit time of dietary residue through the human gut. Gut. Vol. 17, 210-18.
1. Sadik, R., Abrahamsson, H., Stotzer, P.O. (2003). Gender differences in gut transit shown with a newly developed radiological procedure. Scand. J. Gastroenterol., Vol. 38, 36-42.
2. Abrahamsson, H. & Antov, S. (2010). Accuracy in assessment of colonic transit time with particles: how many markers should be used? Neurogastroenterol Motil., Vol. 22, 1164-69.
3. Abrahamsson, H., Antov, S., Bosaeus, I. (2012). Gastrointestinal and colonic transit time evaluated by a single abdominal X-ray in healthy subjects and constipated patients. Scand. J. Gastroenterol., Vol. 23 (suppl. 152), 72-80.
4. Törnblom, H., van Oudenhove, L., Sadik, R., Abrahamsson, H., Tack, J., Simrén, M. (2012). Colonic transit time and IBS symptoms: what’s the link? Am. J. Gastroenterol., Vol. 107(5), 754-60.
5. Abrahamsson, H., Ostlund-Lindqvist, A.M., Nilsson, R., Simrén, M., Gillberg, P.G. (2008). Altered bile acid metabolism patients with constipation-predominant irritable bowel syndrome and functional constipation. Scand J Gastroenterol., Vol. 43(12), 1483-8.
6. Sadik, R., Abrahamsson, H., Ung, K.A., et al. (2004). Accelerating regional bowel transit and overweight shown in idiopathic bile acid malabsorption. Am. J. Gastroenterol., Vol. 99, 711-718.
7. Simrén, M., Bajor, A., Gillberg, PG., Rudling, M., Abrahamsson, H. (2011). Randomised clinical trial: The ileal bile acid transporter inhibitor A3309 vs. placebo in patients with chronic idiopathic constipation-a double-blind study. Aliment Pharmacol Ther., Vol. 34(1), 41-50.
Literature
Keighley, M. & Williams, N. (2018). Keighley & Williams´ Surgery of the Anus, Rectum and Colon. 1664 pages Boca Raton: CRC Press.
Internet Sources
Disney, B., et al. (2019). Cost of Constipation Report. Retrieved January 16, 2023, from https://bowelinterestgroup.co.uk/wp-content/uploads/2019/07/The-Cost-of-Constipation-2019.pdf